Autism Spectrum Disorder, Socio-Affective Deficits, Auditory Sensitivity, Dysregulated Pain Processing, and the Amygdala
By Conner Lee and Jim Stellar
Introduction
Autism Spectrum Disorder (ASD) is a developmental disorder that is shown to affect 1 in 31 U.S. children. ASD is also a heterogeneous condition that encompasses a number of disorders such as Asperger’s and Rett’s syndrome. Due to its multiple clinical forms, ASD pathology is especially difficult to narrow down. However, research indicates ASD most likely develops from genetic and environmental factors that disrupt neuronal organization and cortical development.
As ASD is a neurodevelopmental disorder, analyzing the prenatal and postnatal stage of brain development is important in understanding the origins and progression of such neuro-cortical disruptions. Here, genes such as Reelin appear to play a part in impaired neuronal migration in prenatal brain development. Additionally, the developmental effects of Hepatocyte Growth Factor (HGF) and its receptor MET extend past the prenatal stage and into the postnatal stage which presents a risk for improper motor and sensory development. Environmental factors such as oxidative stress, neuroimmunological dysfunction, and family history of autoimmune disorders can impact the development of ASD in conjunction with the mentioned genetic factors. In terms of treatment, the disorder is potentially modifiable from a clinical perspective, but further research must be done into intervention type, time period of treatment, and other varying factors of the disorder’s pathology.
For this long blog, we focus on the underlying neural mechanisms derived from the genetic and environmental components of ASD in hopes of exploring the neurobiology of autistic individuals. Furthermore, we will focus on cortical and subcortical connections with the limbic system and the amygdala as a number of ASD behavioral symptoms appear to have an underlying emotional aspect. As such, any topics discussed in this paper promote the regulation of the emotional limbic system in an effort to alleviate maladaptive symptoms of ASD rather than the disorder itself. In other words, this paper in no way works to undermine the neurodiverse population but rather grants a deeper dive into brain mechanisms and symptoms originating from ASD related limbic system dysregulation.
ASD as a Neurodevelopmental Limbic System Connection Disorder
Current literature suggests ASD is a multifaceted neurodevelopmental disorder; factors leading to its development include irregularities in normal brain development, neuronal and cortical connectivity, and environmental factors. The variety and interconnected nature of these elements may help explain the previously noted heterogeneous nature of ASD.
We focus on three ASD symptoms related to the limbic system (particularly the amygdala) and its integration with or control by higher cognitive brain systems:
- Socio-affective deficits (1st symptom) could be mediated through connectivity abnormalities from the limbic system to cortical regions. The emotional limbic system’s dysregulation coupled with issues in connectivity could affect higher order functions that manifest as the social deficits seen in ASD. Abnormalities in uncinate fasciculus (UF) development could be a part of the explanation while hypoconnectivity and hyperconnectivity of the amygdala to cortical and subcortical regions of the brain could be another.
- Sensitivity to auditory stimuli (2nd symptom) could be impacted by the dysregulation of typical connectivity development. Cortical regions of the temporal lobe and other brain regions such as the thalamus could be hijacking normal auditory pathways and heightening limbic system activity in response to auditory stimulation. Hypersensitivity to auditory stimuli seems to be a connectivity issue; non-classical auditory pathway dominance overemphasizes limbic system involvement and leads to the amygdala’s exaggerated emotional response to auditory stimuli.
- Dysregulated pain processing (3rd symptom) is an aspect of the broader symptomatology of tactile stimuli processing. Pain was a subsection of interest due to its potential origin in both abnormal limbic system and cortical function. The amygdala could be hyperreactive and hyporeactive in response to pain stimuli. This could help explain the variety in emotional reactions across autistic individuals when exposed to pain. In contrast, the insula, a cortical structure often referred to as the fifth lobe of the brain, could be hyporeactive to pain. The insula plays an integral role in the subjective experience of pain; therefore, an underreactive insula means a lower perception of pain. The interplay of both the amygdala and the insular cortex could influence the dysregulation of pain processing and perception in ASD.
These three symptoms of ASD and their underlying neural qualities will be discussed below.
Symptom 1: Socio-Affective Deficits
Uncinate Fasciculus
The uncinate fasciculus (UF) is a white matter tract connecting the temporal limbic region to the frontal cortex and is believed to impact socio-affective processing. It is hypothesized that this pathway acts as a bridge between emotional processing and higher order cortical processing. Therefore, abnormalities to the UF could be a potential factor in socio-affective and socio-emotional deficits seen in ASD. Interestingly, there seems to be an asymmetrical element to the relationships between the UF and ASD.
The left UF is known as the socio-affective side involved in processes such as the regulation of emotions. In addition, there is growing evidence implicating the abnormal development of the left UF in ASD. Using Diffusor Tensor Imaging (DTI), a number of studies attempt to measure the connectivity and structural integrity of the UF. One such study measures the fractional anisotropy (FA) – a connectivity and structural integrity measurement based on the flow of water molecules – of the left UF in children and adolescents with ASD. It was found that the left UF of the ASD group had lower FA than neurotypical individuals, indicating impaired connectivity of the left UF in ASD. Thus, this could be a possible explanation for the socio-affective deficits of autistic individuals.
However, there is controversy on the use of FA as the sole measure for structural integrity and connectivity. There appears to be variation in the results of FA values in the context of UF measurements in ASD: some studies report higher FA of the UF in ASD while others report lower FA of the UF in ASD. To gain a more complete understanding of the UF in ASD, other DTI measurements such as mean diffusivity (MD), axial diffusivity (AD), and radial diffusivity (RD) should be utilized. Employing such methods reveal curious findings, suggesting UF overgrowth during critical periods of development in children with ASD. UF overgrowth during these integral years of brain development could play a role in socio-affective symptoms. While the controversy behind the UF and its relationship to ASD makes the exact mechanisms difficult to understand, there appears to be a consensus that the proper development of the left UF is vital to socio-affective processing and that some level of UF dysfunction seems to exist in the neurobiology of ASD.
Amygdala and Cortical Connectivity
Social deficits and social anxiety in ASD can be traced back to the limbic system. The amygdala, as the fear center of the brain, garners considerable attention in social anxiety related ASD studies. While previous research explores the amygdala as a whole unit, it is also important to look both deeper into the subunits of the amygdala and expand our view to look at its connections to other brain regions. There is evidence suggesting both hyperconnectivity and hypoconnectivity in the neuroanatomy of ASD individuals. These connections include networks between amygdala subregions and brain regions such as the frontal cortex, nucleus accumbens, and primary motor cortex; specific subregions of examination include the laterobasal subregion (LB), the centromedial subregion (CM), and the superficial subregion (SF).
The laterobasal amygdala is a subregion of interest due to its involvement in social and communication skills. Based on fMRI studies (see the figure below), there seems to be an overall hypoconnectivity from the LB to both cortical and subcortical regions including the inferior frontal gyrus, nucleus accumbens, and anterior cingulate cortex. The inferior frontal gyrus, a frontal lobe structure, works to direct cognitive control. Inhibitions in connectivity to this pathway may suggest an impaired translation from emotional/social intent to cognitive application in the environment. Perhaps this could be an explanation for the social deficits observed in autistic individuals. Furthermore, the reward processing of social situations may be dysregulated in ASD. A hallmark behavioral symptom of ASD is the struggle to create eye contact while socializing. For reasons not clearly understood, ASD adults do not find faces/eyes rewarding to look at starting from a young age. To help explain this phenomenon, abnormal social brain circuitry in hypoconnectivity from the LB to the nucleus accumbens and anterior cingulate cortex (ACC) could be a possible lead. The nucleus accumbens is the center for reward processing in the brain. Lacking connectivity from the LB, the nucleus accumbens may be missing the proper signaling to develop the correlation that facial stimuli is rewarding. The ACC is involved in the broader implications of ASD social anxiety. The hypoconnectivity of LB-ACC interaction potentially mediates the emotional dysregulation in social situations for ASD.
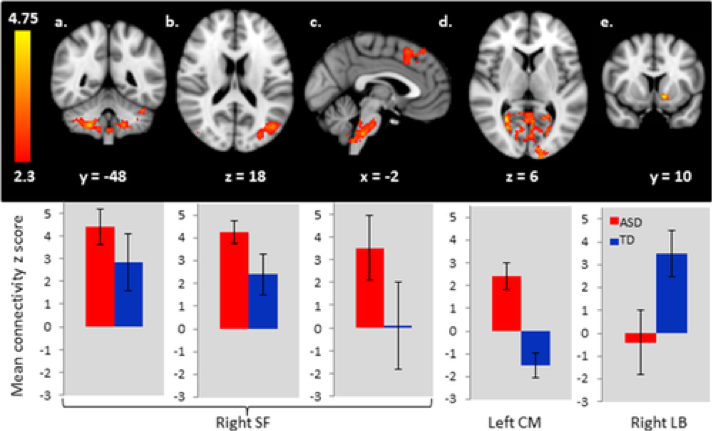
Hypoconnectivity as a modulator for decreased social ability intuitively makes sense (the emotional amygdala fails to signal to higher cortical activity responsible for social behavior). However, the complicated nature of ASD is exemplified through the hyperconnectivity of amygdala subregions to the cortex. When observing the effects of such heightened connections, the symptoms of interest transition to increased sensitivity to stimuli. fMRI studies reveal hyperconnectivity from the CM to primary visual regions and visual association regions. This is reasonable as research indicates that in ASD the brain biases visual information during tasks involving vision and language. In terms of other environmental stimuli, hyperconnectivity from the CM to the brainstem has also been observed in the neurobiology of ASD. Considering the brainstem’s significant function in arousal, the CM’s proliferated connectivity to the brainstem could explain the overall increased autonomic arousal seen in ASD. Anxiety as a whole is most likely mediated by the CM-brainstem pathway, but there could be further implications in the specific social anxiety symptomology of ASD when coupled with both the hypoconnectivity and hyperconnectivity that has been described so far. ASD is a network of connectional irregularities that ultimately manifests in the behaviors of social deficits, social anxiety, and stimulus sensitivity.
Symptom 2: Auditory Sensitivity
Non-Classical Auditory Pathways
Misophonia refers to a condition in which individuals have a disproportionate emotional reaction such as extreme anger and irritation in response to a triggering noise that would not bother a neurotypical individual such as chewing and tapping. It has become increasingly clear that misophonia or hypersensitivity to auditory stimuli has more to do with a dysregulated emotional limbic system rather than a deficit of the auditory system.
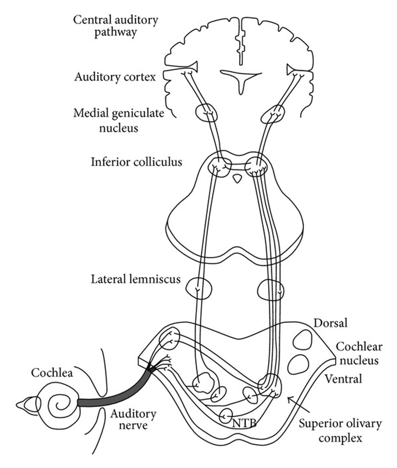
This hypersensitivity to noise is a common symptom of ASD and can be traced back to two different auditory pathways: the classical auditory pathway and the non-classical auditory pathway. In the classical auditory pathway auditory stimuli enters the cochlea where the vibrations are translated in signals that travel down the auditory nerve to the brain stem regions of the pons, superior olivary region, lateral lemniscus, and inferior colliculi. The brain stem then projects to the medial geniculate nucleus of the thalamus and finally to the auditory cortex as shown in the figure below taken from the above reference.
On the other hand, the non-classical auditory pathway deviates from the classical auditory pathway and projects to the limbic system (see the figure below). More specifically, the deviation occurs at the lateral lemniscus of the brainstem. The lateral lemniscus communicates to and receives information from the reticular formation, which then projects into the thalamus. The thalamus connects to the auditory association cortex which in turn projects to the limbic system. Furthermore, separate projections from the thalamus connect directly to the amygdala adding another layer of limbic system connectivity. In neurotypical individuals, the nonclassical auditory pathway dominates in childhood until the age of 8 in which the classical auditory pathway takes over. In autistic individuals, however, it seems that the non-classical auditory pathway continues to dominate past the age of 8 which could serve as a possible explanation for misophonia in ASD. The continued involvement of the limbic system and the amygdala as a major part of auditory processing leads to exaggerated emotional reactivity in response to auditory stimulation.
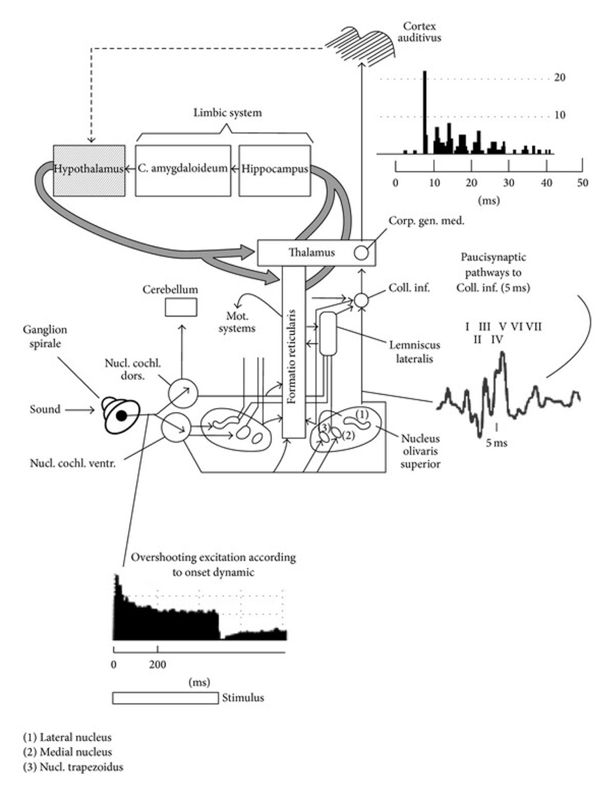
Another important aspect of the non-classical auditory pathways is the integration of somatosensory stimulation namely touch. In neurotypical children, using the non-classical auditory pathway, touch may be a soothing stimuli in response to fearful auditory stimuli. However, in autistic children it may be that both auditory stimuli and somatosensory stimuli appraisals are negative, impairing their ability to associate certain sounds as non-threatening.
The Vagus Nerve and Fight or Flight
Fight or flight behaviors can be encompassed by the term negative emotional behavioral reactions. Negative emotional behavioral reactions are enacted in response to distressing stimuli and situations. In the context of ASD, negative emotional behavioral reactions can be the product of misophonia. We discussed the non-classical pathway and its role in connecting the auditory systems of the brain to the limbic system. In looking at the actual behaviors mediated by misophonia, the vagus nerve’s connection from the limbic system to the periphery and the autonomic nervous system seems to be a promising area of research.
Porges’s Polyvagal Theory (also see) posits that fight or flight responses are mediated by emotions and the periphery. The vagus nerve integrates emotional information from the limbic system and innervates areas of the periphery responsible for stress response. For example, if a noise from the environment is appraised as distressing, the vagus nerve utilizes that information to increase heart rate. Increased arousal subsequently leads to the behavioral effects of fight or flight.
In ASD, the vagus nerve is not inherently overactive in relaying limbic system signaling to the autonomic nervous system. Instead it acts as the mechanism connecting emotional reactivity to actual ASD behaviors such as increased anxiety behaviors, shutting down, and babbling in response to certain auditory stimuli.
Symptom 3: Dysregulated Pain Processing
Pain Processing and Perception
Pain processing and perception in ASD is believed to follow an atypical top down regulation pattern. Pain processing can be split into two parts: the nociceptive matrix and the salience matrix as shown in the figure below from the reference cited above.
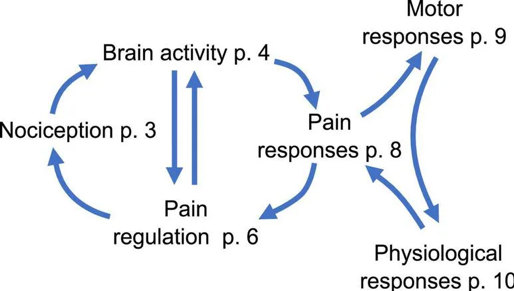
Nociception, or pain stimuli detection, is the first step involved in processing pain. A key player in this step is the thalamus and its connection to cortical and subcortical structures. In ASD, both hypoconnectitvity and hyperconnectivity was seen between the thalamus and cortical areas, and hyperconnectivity was observed between the thalamus and the amygdala. The heterogeneity in connectivity between cortical regions and the thalamus reveals dysregulation in the cortex’s inhibitive quality to sensory information such as pain. This dysregulation paired with the overall increased connection from the thalamus’s sensory integration to the amygdala’s emotional reactivity could explain the exaggerated emotional response to non-painful stimuli in ASD (allodynia) as shown in the figure below taken from the reference cited above.
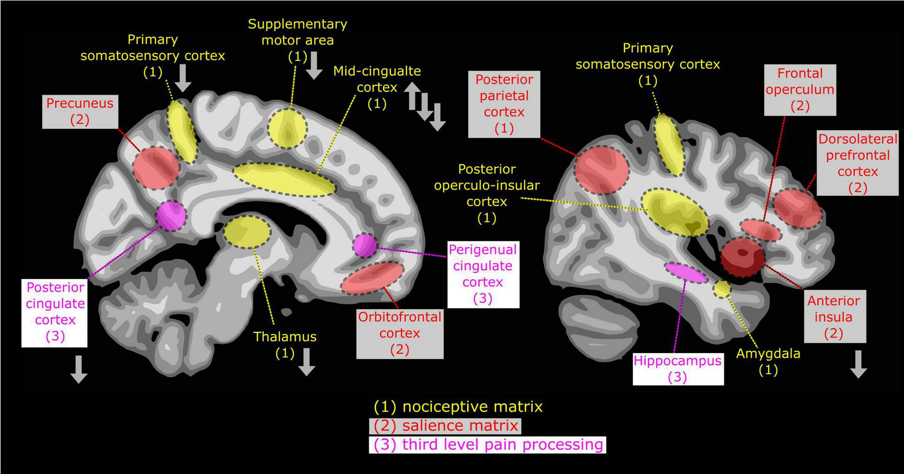
The salience matrix brings attention to painful stimuli (or any stimuli that reaches a specific threshold). For the conversation of this element of pain processing, we focus on the insula and the hippocampus-prefrontal cortex-amygdala network. As mentioned, the insula is a brain region located in the lateral sulcus separating the frontal and parietal lobes of the brain. It is a brain structure cited to be involved in the perception and encoding of pain as the brain’s center for interoception. This means that the insula plays a key role in bodily self awareness. In ASD, the insula seems to have decreased activation in comparison to neurotypical individuals. This decrease could be correlated to the abnormalities in the insula’s structure, function, and connectivity. Furthermore, suppression of the insula could help explain abnormal pain recognition patterns – both hyporeactive and hyperreactive – in ASD.
Another part of the salience matrix is the hippocampus-prefrontal cortex-amygdala network. The prefrontal cortex (PFC) is involved in top down control over pain processing. There seems to be a form of PFC dysregulation in ASD as studies reveal both decreased PFC activation overall and decreased PFC responses to pain stimuli. Lack of top down control from the PFC affects the downstream aspects of hippocampal and amygdala activation in response to pain which seems to affect neuroplasticity and memory impairments related to pain. Based on these findings, the neurobiology of ASD could deviate from neurotypical individuals through the abnormal rewiring of pain pathways.
The top down model of pain processing follows the connections between the PFC, thalamus, periaqueductal gray (PAG), and the amygdala. The amygdala is uniquely involved in the perception of pain as it can label a pain experience with emotional value. The appraisals the amygdala makes towards pain seems to depend on the frequency of experiencing such pain stimuli. Acute pain derived amygdala activation leads to hypoalgesia or decreased sensitivity towards pain. This makes sense from an evolutionary stand point as decreased sensitivity to pain in dangerous situations most likely would increase survival chances. In contrast, chronic pain derived amygdala activation leads to hyperalgesia, or an increased reactivity towards pain. As autistic individuals show heterogeneity in pain reactions, perhaps the amygdala is responsible for modulating the expression of both hypoalgesia and hyperalgesia and can also help researchers understand why such heterogeneity in reaction types exist. Furthermore, there is growing evidence that weak PFC connectivity with the amygdala plays a role in less emotional reaction to pain. This suggests that the interplay of cortical control and amygdala reactivity is a key aspect of impaired pain processing.
Conclusion
From this limited survey we were able to focus on the limbic system and the amygdala as a driving force in ASD. Particularly, the amygdala and its connectivity to cortical and subcortical regions, seem to modulate core ASD symptoms such as socio-affective deficits, sensitivity to auditory stimuli, and dysregulated pain processing. ASD is a complicated developmental disorder characterized by its heterogeneity across autistic individuals. We hope this paper helped clarify some potential sources for such diversity in behavioral symptomology and inspire future work that delves deeper into the
